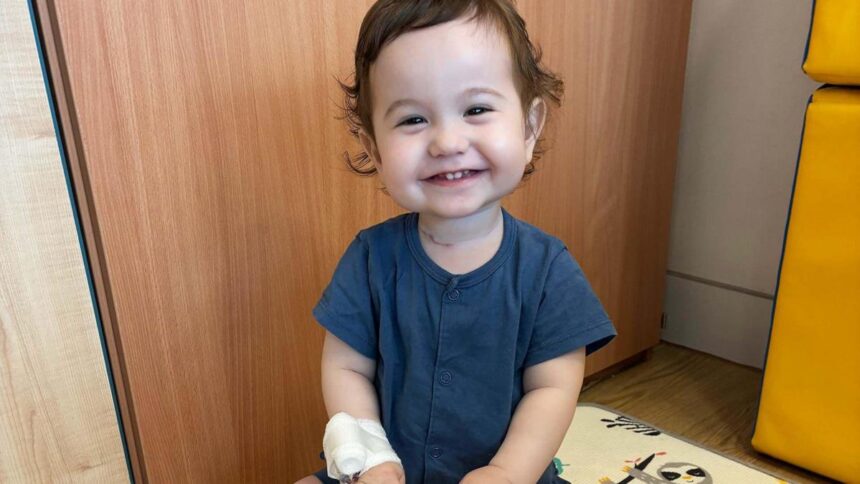
In a remarkable advancement for paediatric healthcare, UK researchers have successfully developed functioning oesophageal tissue in laboratory settings, paving the way for potential transplants in young patients with congenital conditions. This breakthrough, detailed in the esteemed journal Nature Biotechnology, represents a beacon of hope for children like two-year-old Casey Mcintyre, who was born with a significant portion of his oesophagus missing.
A New Dawn for Casey and Others
Casey Mcintyre was born with a staggering 11 centimetres missing from his oesophagus, a condition that has necessitated multiple surgeries since his birth. His mother, Silviya, recalls the anxious anticipation before his arrival, knowing he would face serious challenges related to his food pipe. To date, doctors have repositioned his stomach to compensate for the absence of oesophageal tissue, but Casey still relies on a feeding tube as he works toward mastering the art of swallowing.
Silviya shared, “The repeated surgeries have left him with some damage to his vocal cords, so he’s currently catching up with his speech and noises. Once he can consume enough food orally, we’ll be able to remove the tube.” Casey’s father, Sean, reflected on the unexpected lessons parenthood has taught them, from managing tube feeds to navigating urgent hospital communications in the dead of night. He stated, “To look at him, he’s just amazing, and we are incredibly proud of him. Whatever the medical team has done feels miraculous. The prospect of a single operation that could provide a functioning oesophagus would truly be life-changing.”
It is estimated that approximately 18 infants in the UK are born each year with similar oesophageal defects, making this research particularly significant.
Groundbreaking Research in Animal Models
The pioneering study involved creating lab-grown oesophageal tissue using Göttingen minipigs, a breed selected for its anatomical and physiological similarities to human children. The research team, led by Professor Paolo De Coppi at Great Ormond Street Hospital and University College London, took an existing pig oesophagus, stripped it of its cells while preserving its structural framework, and infused it with new cells. This engineered tissue was then placed in a bioreactor, a specialised environment designed to nurture cellular growth, for a week.
Eight minipigs received the transplants, with five surviving through to the six-month mark, exhibiting fully functional grafts with muscle, nerve, and blood vessel integration. The researchers found that the pigs developed effective swallowing muscles, essential for moving food towards the stomach. Remarkably, no anti-rejection medications were needed, as the implants utilised the animals’ own cells.
Looking Ahead: A Future of Hope
Professor De Coppi expressed optimism about the potential to translate this groundbreaking research into clinical treatments for children within a five-year timeframe. He noted, “The oesophagus is a complex organ without a dedicated blood supply, so it cannot be transplanted in a conventional sense. Developing alternatives requires animal models that closely mimic human anatomy and function.”
However, it is important to note that this innovative graft would not be suitable for adult patients suffering from oesophageal conditions such as cancer, as it is specifically designed to grow with children as they develop.
Why it Matters
The development of lab-grown oesophageal tissue holds profound implications for paediatric medicine, particularly for those grappling with congenital anomalies. For families like Casey’s, this research signifies not just a scientific achievement, but a potential end to the arduous cycle of surgeries and uncertainties that often accompany such conditions. As scientists continue to advance this promising technology, the dream of improved quality of life for young patients may soon become a tangible reality, offering hope to countless families facing similar challenges.